DCC Compliance Testing
Cannabis products intended for medical or recreational sales must pass the CA Compliance analysis conducted by a licensed, third-party analytical lab.
Compliance regulations are extensive and require close attention to detail to ensure passing results. From packaging and labeling to quality assurance testing, cannabis organizations must follow these regulations to a tee to prevent any product recalls or failures. The average turnaround time for DCC compliance testing is 4 business days, with expedited options available. For more information or to schedule a compliance pick-up please reach out to an account manager. InfiniteCAL’s team of experts is here to help guide you through the process to get high quality products to market.


Am I Ready for Compliance?

Sample Requirements

TAT & Results

Useful Info
How you consume &
What is required
Smokables

Flower
- Cannabinoid Potency
- Pesticides
- Microbe Inhalable
- Heavy Metals
- Mycotoxins
- Water Activity
- Foreign Material
Pre-Rolls
- Cannabinoid Potency
- Pesticides
- Microbe Inhalable
- Residual Solvents
- Heavy Metals
- Mycotoxins
- Water Activity
- Foreign Material
Concentrates
- Cannabinoid Potency
- Pesticides
- Microbe Inhalable
- Heavy Metals
- Mycotoxins
- Water Activity
- Foreign Material
Edibles

Edibles
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Water Activity
- Foreign Material
Beverages
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Water Activity
- Foreign Material
Other

Tinctures
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Foreign Material
Topicals
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Foreign Material
Capsules
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Foreign Material
Sublingual Strips
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Foreign Material
Bath Bombs
- Cannabinoid Potency
- Pesticides
- Microbe Non-Inhalable
- Residual Solvent
- Heavy Metals
- Mycotoxins
- Foreign Material
Move over potency
It’s all about
the Terps
Why terpenes matter and why you should test for them in your cannabis and hemp products.
To understand the differences between different cultivars and their effect on our bodies, we have to move beyond looking at the THC content and start thinking about the other components which make each strain unique. Terpenes are tiny molecules with powerhouse effects that have a huge impact on the character of your cannabis. In any cannabinoid product, terpenes and cannabinoids interact synergistically to create an entourage effect.


Am I ready for Compliance?
How to prepare for your next pick-up in California
The Paperwork
3 things we need:
-Prep For Sample Collection Form
-Distribution or Microbusiness License
-Producer License for each batch
Please send licensing info to [email protected]
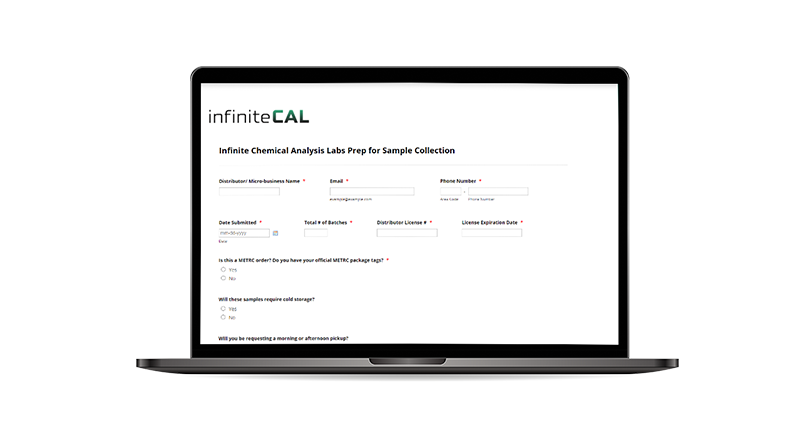
When submitting your
Prep for Sample Collection Form
• Select “YES” if you are submitting Metrc samples and have your official Metrc package tags.

• “Sample Name” must match the “Item Name” on your Metrc manifest.

• If you’d like the source package tag included on the COA, please include it in the “Notes” section of the Prep Form and MUST be labeled on the product at time of sampling.
• Also, please provide a breakdown of different sizes and if you need any special temperature requirements.
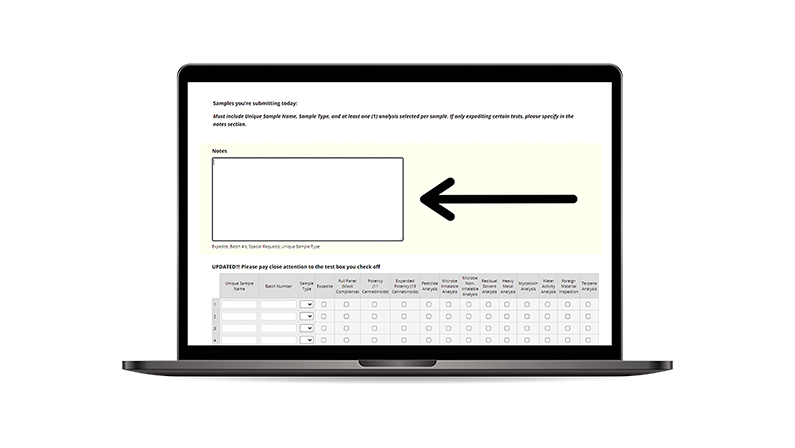
• Assign a Tag to the test sample package.
• Provide the whole physical tag to the lab’s Sample Technician.
• The tech will affix the top portion of the tag to the physical bag holding the representative sample.
• The bottom portion will be used on your official Chain of Custody form to accompany the transporter manifest.

• Every container that your product is in must be in final form, consumer-facing packaging prior to the compliance sampling event.*
*Expection for bulk flower

On 
• You must select ![]() in order for the lab to accept your manifest.
in order for the lab to accept your manifest.
• A teardrop icon will appear once the manifest has been properly submitted.
• One test sample package should be created from a package, even if there are multiple package sizes. In this case, submit as a multi-package.
• Once your Manifest is transferred to the lab, you have 24 hours to make any edits or changes.

Transferring Test Samples to the Lab
• Total Batch Size on your Prep for Sample Form must equal the total amount of the entire batch.
• Total batch size for Flower should always be notated in grams whether it is packaged or unpackaged.
(NOTE: Batches of flower cannot exceed 50 lbs, other product types cannot exceed 150,000 units)
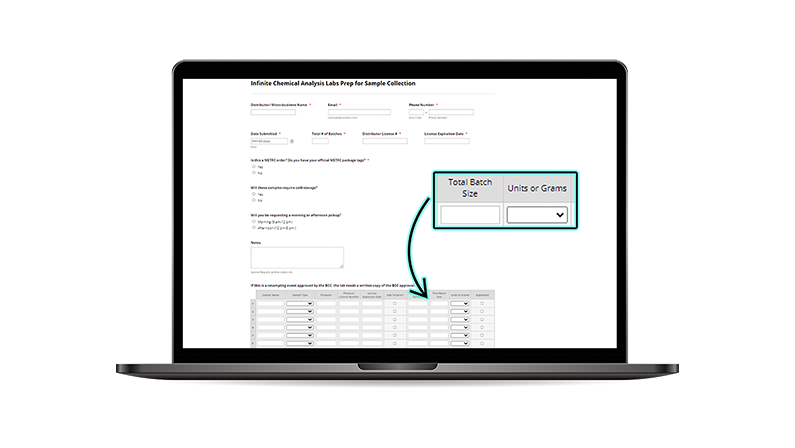

• Quantity shipped will be a representitve sample of the entire batch according to DCC regulations and Total Batch size on your Prep for Sample Form.
• Any chemical variation to a product (added terpenes,flavors, ect.) requires a separate batch number and therefore separate batch testing.
Please verify the quantity of samples taken with the sample technician or InfiniteCAL’s scheduling director, before transferring your Metrc manifest.
Compliance Sampling
When our Sample Tech arrives make sure all Products are:
• Separated
• Quarantined
• Accessible
• Accurately Labeled
with the info from your Prep For Sample Collection Form
What is a compliant batch?
The following table displays sample minimums for compliance testing as required by the DCC.
However, lab minimums may not be the same as the DCC’s (see table).
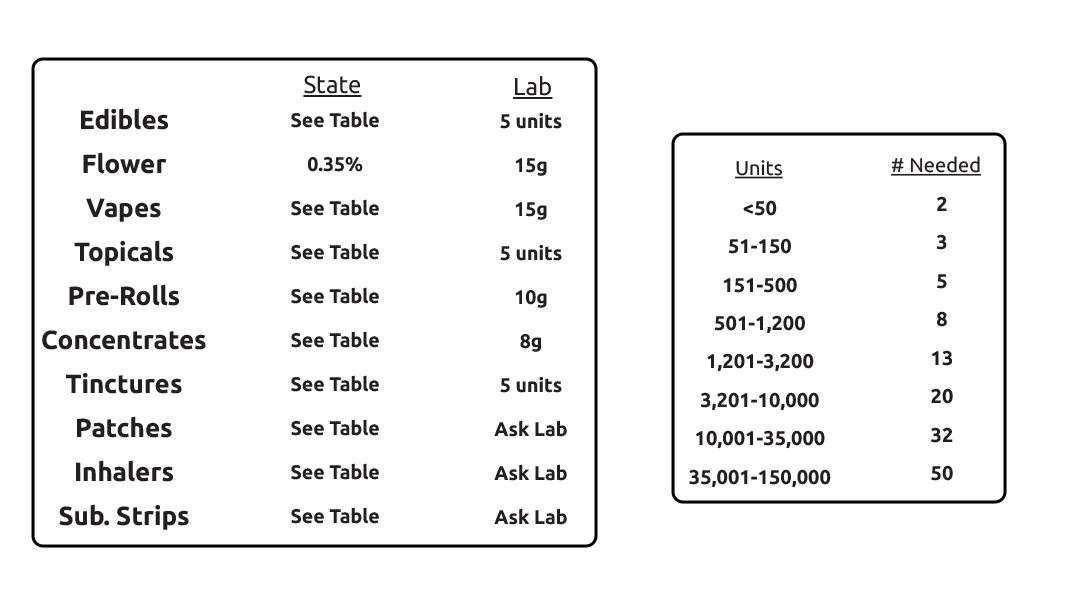
If you have any questions concerning sample minimums or would like to know how many units are to be taken from your batch prior to pick up, please contact the lab.
Results
Results are available wherever you are. InfiniteCAL uses Confident Cannabis, a third-party reporting platform that allows 24/7 access to sample results, unique QR codes, features for sharing COA’s with buyers and analytical data mining.
Turnaround Time
InfiniteCAL’s average turnaround time for compliance is 4 business days with expedited options to get results as quickly as 2 business days.

- All manufactured products and pre-rolls must be sampled in its final, consumer-facing packaging including a batch number, Metrc UID, manufacturing and packaging date, product identity, net weight or volume, and the California Universal Symbol.
- All products must have a Tamper Evident Sticker and Child-Resistant Packaging.
- Cannabis edibles must include the words “Cannabis-Infused” on the primary panel of the packaging.
- Cannabis packaging should not be attractive to children.
- Once samples are picked up, the laboratory must complete the compliance analysis and report the results to the Department of Cannabis Control.
- There is no process for the licensee requesting testing to be canceled once the samples have been collected from the distributor’s premises.
- The Department of Cannabis Control’s regulations do not permit the reporting of any results prior to the release of the regulatory compliance COA.
- We cannot amend any COA after issuance; if a product fails compliance, the licensee has up to 30 days to submit a corrective plan of action to the State Department of Public Health or the Bureau.
- If you need to request to remediate a compliance batch please email
- If you need to request a resampling please email