Quality Assurance
Quality Assurance – QA – is the process of verifying whether a product meets required specifications. Many companies use QA testing to gain insight on their current products or to create new systems that will help them maximize their intended goals. Most often, the goal is to prepare a product for State Compliance Testing. QA testing is an advantageous asset in a compliance-regulated industry, which is why we offer the same tests needed at the compliance level a la carte. Clients can pick and choose different tests individually from our menu depending on your testing focus.


Submitting Samples

Sample Requirements

TAT & Results

Useful Info
Our Testing Services

Cannabinoid Profile
Our cannabinoid potency test provides quantitative information on the active cannabinoids present in a sample.

Residual Solvents
There are dozens of different types of cannabis products, and a number of them require the use of one or more processing solvents during production.

Heavy Metals
Heavy metals are associated with serious adverse health effects in humans, ranging from birth defects to cancer.

Vitamin E Acetate
Vitamin E Acetate Test can determine the presence of this potentially harmful additive in vape products.

Terpenes
Terpenes are naturally existing small organic molecules produced by a wide variety of plants, including cannabis.

Microbial
Microbial Testing is a vital component when it comes to ensuring any major consumer product is free of bacteria and mold that could potentially cause illness.

Water Activity
The measurement of water activity is a key parameter in the quality control of moisture-sensitive products or materials.

Microextraction
Our high-throughput service allows for rapid screening of extracts allowing you to quickly determine which biomass is best for you.

Pesticides
Because Pesticides are used in the cultivation of cannabis and hemp, they need to be tested for in every product.

Mycotoxins
One of the most prominent dangers to consumers can come in the form of mycotoxins found on or in the cannabis plant and its extracts.

Foreign Materials
The state requires that all cannabis samples be thoroughly inspected using a microscope and visual inspection.

Sampling Event
We offer Trim and Oil sampling events. Our objective as a third-party lab is to assist in an unbiased R&D sampling.
Additional Services
We Offer

Compliance Testing
InfiniteCAL is a fully licensed DCC and ISO/IEC 17025 accredited cannabis testing lab. Regulatory compliance testing is a state-mandated requirement for all cannabis products sold in the State of California.

California Hemp Testing
If your company is registered to grow Industrial Hemp in the state of California, the CDFA requires samples from your crop(s) to be analyzed 30 days before harvest to determine total Tetrahydrocannabinol levels do not exceed 0.3%. InfiniteCAL is certified to conduct the official CDFA analysis and works closely with the local CDFA county offices and registrants to ensure compliance.

Colorado Hemp Testing
The Colorado Hemp industry can now count on InfiniteCAL for accurate, reliable, and constant lab results for their compliance testing, as well as any quality assurance testing.
We accept drop-off samples M-F 8 am-7 pm at our lab located in the Miramar area of San Diego and samples sent via FedEx and UPS.
Address:
Infinite Chemical Analysis
8312 Miramar Mall
San Diego, CA 92121
We can also schedule a pickup for locations across California. To schedule a pickup, please get in touch with your account manager or contact the lab directly.
858 623-2740
When samples are dropped off, picked up, or shipped, please fill out and include a QA Chain of Custody so we know precisely whose samples they are and what tests to perform.
Each product requires a different amount of sample per QA test. General guidelines are listed as follows:


These are general sample minimums. Science is not always perfect, and we may need to request more to verify results. If you feel comfortable providing more sample material than requested, we will not decline. InfiniteCAL recommends always providing a representative sample of the entire harvest or production batch
Results
Results are available wherever you are. Our third-party reporting platform allows 24/7 access to your sample results, unique QR codes, features for sharing COA’s with buyers, and analytical data mining.
Turnaround Time
Our turnaround for QA testing is 2-3 days, which we meet or exceed with a 97% success rate. Expedited options are available for those who need their results by the next business day.

Useful Info
- QA testing may be requested by anyone 21+.
- QA tests can be ordered at any time without the official guidelines. Official Licenses are not required for QA testing.
- Results from QA testing cannot be used to legally sell products to storefronts, but are a good way to know what the status of your products are before submitting them for official tests and can decrease the fail rate for your products.
Cannabinoid Potency Testing
The term “cannabinoid” originally referred to the class of biologically active compounds that occur naturally in Cannabis sativa. The commonalities between these compounds were: a central aromatic ring, two ortho-oxygen atoms on the aromatic ring, and a total of 21 carbons. Now the term takes on a broader definition to include two different subgroups: (1) phytocannabinoids – the compounds that naturally occur in Cannabis sativa – and (2) synthetic cannabinoids – novel compounds that are synthesized in a lab and bear structural similarity to phytocannabinoids.
Our potency test provides quantitative information on up to 19 cannabinoids, including phytocannabinoids and synthetic cannabinoids.
Cannabinoids we test for:
Standard Cannabinoid Profile (11 Cannabinoids) Delta-9-Tetrahydrocannabinol, Tetrahydrocannabinol-Acid, Cannabidiol, Cannabidiol-Acid, Cannabinol, Cannabigerol, Cannabigerol-Acid, Delta-8-Tetrahydrocannabinol, Tetrahydrocannabivarin, Cannabichromene, Cannabidivarin
Expanded Cannabinoid Profile (19 Cannabinoids): Standard Cannabinoid Profile + Cannabichromene-Acid, Cannabichromevarin, Cannabidivarin-Acid, Cannabicyclol, Cannabicyclol-Acid, Cannabinol-Acid, Cannabitriol, Tetrahydrocannabivarin-Acid.
How We Test For Cannabinoids
We test for cannabinoids using Ultra High-Performance Liquid Chromatography with a Diode Array Detector (UHPLC-DAD). Chromatography separates the cannabinoids and other organic molecules based on interactions with the stationary and mobile phases as they travel through a column. Once adequate separation is achieved, the cannabinoids are quantified by the diode array detector which measures the absorbance of UV-Visible light exhibited by each compound. The concentration of each cannabinoid can be determined from the intensity of absorption. For more information about cannabinoid potency testing, please refer to pg. 113-114 of the DCC Regulations.
Moisture Content
The amount of moisture present in Cannabis plant material directly affects potency levels. Therefore, the DCC requires labs to adjust for moisture content using the following equation:
Dry-weight percent cannabinoid = wet-weight percent cannabinoid / (1 − percent moisture / 100)
Terpenes influence the aroma and flavor of these plants, including cannabis. Terpenes have also been purported to enhance the experience and therapeutic benefits when combined with cannabinoids.
How We Test For Terpenes
We test for the most common cannabis terpenes using Headspace Gas Chromatography-Mass Spectrometry (HS-GCMS), enabling us to extract and identify each terpene found in a sample. For more information about terpene testing, please refer to pg. 114 of the DCC Regulations.
Terpenes We Currently Test For:
- α-Bisabolol
- Camphene
- δ-3-Carene
- β-Caryophyllene
- Caryophyllene Oxide
- p-Cymene
- Eucalyptol
- Geraniol
- (-)-Guaiol
- α-Humulene
- δ-Limonene
- Linalool
- β-Myrcene
- cis-Nerolidol
- trans-Nerolidol
- α-Pinene
- β-Pinene
- α-Terpinene
- γ-Terpinene
- Terpinolene
- α-Cedrene
- α-Terpineol
- Citronellol
- β-Eudesmol
- Borneol
- Camphor
- Cedrol
- Fenchol
- Fenchone
- Geranyl Acetate
- Isoborneol
- Menthol
- Pulegone
Consumer safety is our number one concern. The DCC requires that all cannabis products sold for consumption be screened for both Category 1 & 2 pesticides. A product can not have any amount of a Category 1 pesticide, and can have only trace amounts of Category 2 pesticides.
How We Screen For Pesticides
We use Liquid Chromatography- Mass Spectrometry (LCMS) and Gas Chromatography- Mass Spectrometry (GCMS) to analyze the required 66 pesticide levels within a sample. For more information on pesticide testing, including the action level of each analyte tested, please refer to pg. 107-110 of the DCC Regulations.
Pesticides We Screen For:
Category 1 Pesticides
- Aldicarb
- Carbofuran
- Chlordane
- Chlorfenapyr
- Chlorpyrifos
- Coumaphos
- Daminozide
- DDVP (dichlorvos)
- Dimethoate
- Ethoprop(hos)
- Etofenprox
- Fenoxycarb
- Fipronil
- Imazalil
- Methiocarb
- Methyl Parathion
- Mevinphos
- Paclobutrazol
- Propoxur
- Spiroxamine
- Thiacloprid
Category 2 Pesticides
- Abamectin
- Acephate
- Acequinocyl
- Acetamiprid
- Azoxystrobin
- Bifenazate
- Bifenthrin
- Boscalid
- Captan
- Carbaryl
- Chlorantraniliprole
- Clofentezine
- Cyfluthrin
- Cypermethrin
- Diazinon
- Dimethomorph
- Etoxazole
- Fenhexamid
- Fenpyroximate
- Flonicamid
- Fludioxonil
- Hexythiazox
Category 2 Pesticides Cont
- Imidacloprid
- Kresoxim-methyl
- Malathion
- Metalaxyl
- Methomyl
- Myclobutanil
- Naled
- Oxamyl
- Pentachloronitro benzene
- Permethrin
- Phosmet
- Piperonyl Butoxide
- Prallethrin
- Propiconazole
- Pyrethrins
- Pyridaben
- Spinetoram
- Spinosad
- Spiromesifen
- Spirotetramat
- Tebuconazole
- Thiamethoxam
- Trifloxystrobin
While generally removed during processing, the residual solvents remaining in cannabis products can be harmful when ingested or inhaled. Therefore, residual solvent screening is the only way to ensure that these products are safe for consumption.
How We Test For Residual Solvents
We use Headspace Gas Chromatography- Mass Spectrometry (HS-GC-MS) to quantify the 20 solvents required by the DCC. For more information on residual solvent testing, including the action level of each analyte, please refer to pg. 106-107 of the DCC Regulations.
We perform qPCR microbial analyses for inhalable and non-inhalable products and have options for 3M plating for different types of bacteria.
How We Test For Microbial Growth
We use two different methods to test for microbial growth:
The first method is plating, which allows us to identify the total colony-forming units per gram of sample. We can then estimate the total bacterial cell concentration within a sample. This method can test for total aerobic bacteria, yeast and mold, STEC E. coli, and coliforms.
The second method involves quantitative polymerase chain reactions (qPCR) to determine the presence of harmful microbes within a sample. The process begins by adding a nutrient-rich broth to the sample, making it the perfect living condition for any microbes to grow after an incubation period. For example, salmonella and Shiga toxin-producing E. coli (STEC) require a minimum of 18 hours incubation, while the mold, Aspergillus, needs at least 24 hours of incubation before testing can begin. These extendked incubation periods give the microbes (if present) adequate time to grow and replicate to produce enough DNA for our instrument to detect. After the samples are brought out of the incubator, we use validated methods to begin DNA extraction from each sample. After DNA extraction is complete, each sample is set onto a plate that the qPCR instrument will read and use fluorescence to verify the presence of an analyte.
We are using Medicinal Genomic’s Multiplex Detection Assay to detect hop latent viroid, lettuce chlorosis virus, and cannabis cryptic virus in cannabis and hemp plants.
This assay tests based on gene amplification and detection by reverse-transcriptase quantitative PCR. Medicinal Genomics says the virus species will be read on the following fluorophores:
- Cannabis DNA – HEX
- Hop Latent Viroid – FAM
- Lettuce Chlorosis Virus – ROX
- Cannabis Cryptic Virus – Cy5
The kit comes with a Leaf Lysis Solution, a Master Mix solution, and a positive control virus multiplex.
Our sample minimum is five fan leaves from various portions of the plant. Each set of leaves should be sealed in Ziploc bags and labeled with a unique identifier.
We Offer Custom Microbial Plating For:
- Aerobic Bacteria (APC)
- Coliform
- E. Coli
- Yeast & Mold
We also offer pH testing for samples already in aqueous solution.
Mycotoxins are a secondary metabolite produced by fungi and some molds that readily colonize crops and can cause major health issues for consumers.
How We Test For Mycotoxins
We use Liquid Chromatography with tandem Mass Spectrometry (LC-MS/MS) to test for four Aflatoxins and Ochratoxin A required by the DCC. For more information regarding mycotoxin testing, including the action level of each analyte tested, please refer to pg. 111 of the DCC Regulations.
Mycotoxins We Test For:
- Aflatoxin B1
- Aflatoxin B2
- Aflatoxin G1
- Aflatoxin G2
- Ochratoxin A
Cannabis plants are more susceptible to heavy metal contamination because they are in a unique class of hyperaccumulating plants that can tolerate uptake of significant levels of heavy metals from soil before their growth cycles are negatively affected. Thus heavy metal testing is vital to ensure consumer safety.
How We Test For Heavy Metals
ICAL uses a state-of-the-art technique called Inductively-Coupled Plasma-Mass Spectrometry (ICP-MS) to quantify these metals. ICP-MS is a highly sensitive technique that detects analytes down to the Part Per Trillion (ppt) range. Typically, a sample is fully digested in a microwave apparatus, diluted, and then injected into the ICP-MS instrument. The sample is then ionized in an inductively coupled plasma flame that reaches temperatures between 6,000K and 10,000K (comparable to temperatures on the Sun!). The resulting ions are separated and quantified by their mass-to-charge ratios (m/z) in a mass spectrometer. For more information on heavy metal testing, including the action level of each analyte tested, please refer to pg. 112 of the DCC Regulations. Current DCC regulations require quantification of arsenic, cadmium, mercury, and lead (referred to as the “big four”) in all cannabis and cannabis-derived products. We also have developed methods for quantification of copper, nickel, and chromium.
Heavy Metals We Test For:
- Cadmium
- Lead
- Arsenic
- Mercury
- Copper (by request)
- Nickel (by request)
- Chromium (by request)
If there is too much water in a product, there is a risk of microbial growth and water migrations. This can lead to clumping, changes in consistency, and reduced shelf-life.
How We Test For Water Activity
We use a water activity probe to measure water activity for solid edible and packaged dried flower products as required by the DCC. Water activity is expressed as a decimal to reflect the ratio between the vapor pressure of the consumable itself, when in a completely undisturbed balance with the surrounding air media, and the vapor pressure of distilled water under identical conditions. For more information on water activity testing, including action levels, please refer to pg. 105-106 of the DCC Regulations.
Foreign Material Inspection
This can include hair, insects, excreta, or any related adulterant that may be hazardous or cause illness or injury to the consumer. For more information on foreign material inspections, please refer to pg. 111-112 of the DCC Regulations.
Vitamin E Acetate is a popular diluent & thickener commonly found in illicit vape products. While Vitamin E acetate has been used in dietary supplementation and in the cosmetics industry for years, inhaling the lipid can cause serious lung injury. While there are currently no California regulations regarding testing for the diluent, we highly recommend running this test when sourcing your distillate to protect your consumers and enterprise.
How We Test For Vitamin E Acetate
We’ve established a fully validated method to test for vitamin E acetate using Liquid Chromatography Triple Quadrupole Mass Spectrometry (LC-MS/MS).
Guidelines for a Sampling Event
When our Sample Tech arrives at your facility, your trim or your oil (must be heated between 50-60°C*), ready to go, separated & accessible!
*The purpose of heating the oil is to decrease the viscosity and is an essential part of homogenizing oil properly. It is important to not overheat and not expose the oil to air to prevent degradation and or oxidation.
Before the Pickup
Prior to the sampling, we will need to know the Total Batch Size. This is the number of bags/bins the trim is in and the size of those bags/bins or the number of jars the oil is in and the size of those jars.
Examples:


How much will we need?
• Your total batch size
• Tests requested
• Sampling size of your preference or company SOP (must meet ICAL’s lab minimum requirements, please contact the lab for details.)
ICAL’s Sampling Suggestions
To ensure a representative sample of your entire batch is taken we recommend pulling:
80 grams for every 50 pounds of trim.**
8-10 grams for every one kilogram of oil.**
**80 grams of flower per 50 pounds of trim or 8-10 grams for every one kilogram of oil is ICAL’s suggestion based on internal SOP’s. It is only required to provide the minimum sample size for each specific analysis ran. However, to receive the most accurate results, it’s essential for InfiniteCAL to analyze a fully homogeneous, representative sample of an entire batch of products. Lack of homogeneity or too small a sample size may lead to inconsistent results.
Schedule your sampling today!
Email: [email protected]
Call: 858 623-2740
Pesticides that may be present at very low levels in biomass can concentrate during the extraction process causing elevated levels to show in the extract. Therefore, it is important to screen biomass on a small scale to help determine which lots are suitable for large scale extraction.
Our high-throughput microextraction service allows for rapid screening of extracts from biomass allowing you to quickly determine which biomass is best for your process.
It is crucial that the sample you provide to us is representative of the entire lot of biomass. We recommend providing a 100 g sample for every 100 lbs of biomass.
EX:
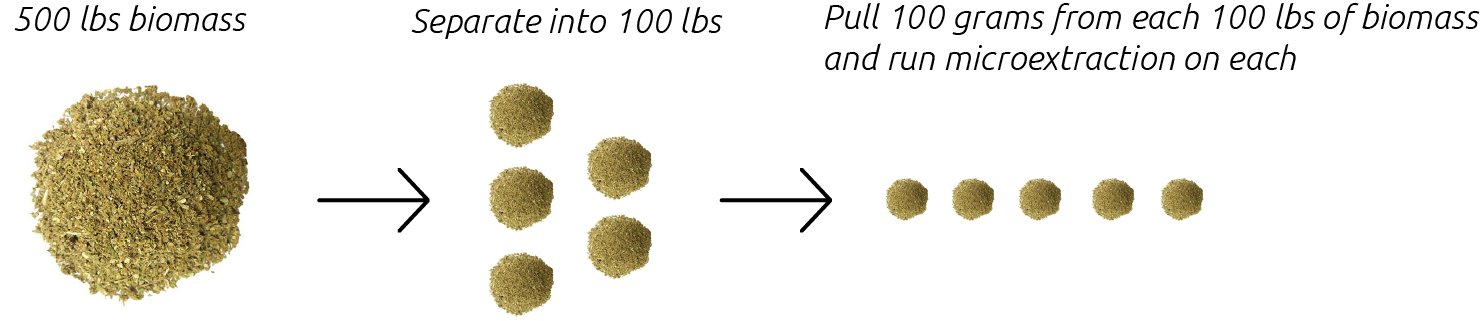
When sampling the 100 g of material, we advise that you take 1-2 g portions from as many different locations within the lot as possible (top/middle/bottom, left/right, etc).
The expected standard turnaround time for the microextraction service and subsequent pesticide analysis is 4-5 business days.
Disclaimer: InfiniteCAL does not provide any guarantee that results from microextractions will match your in-house extraction. Pesticides may extract and/or degrade at different rates under different extraction conditions. We will always do our best to provide you with useful information so you can make informed decisions, however InfiniteCAL cannot be responsible if the pesticide profile determined from microextraction does not match exactly with the pesticide profile from the full in-house extraction. If needed, our lab managers can work with you closely to develop custom conditions that will resemble your in-house extraction.










